December 2018 – The CenterWatch Monthly : pdf
$79.00
Product Details
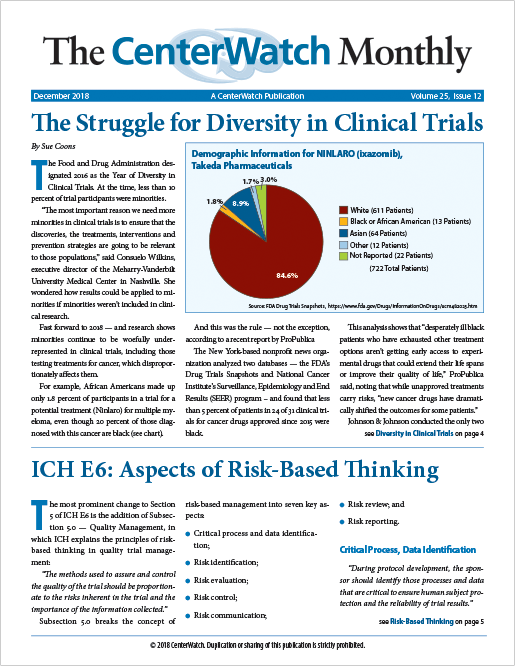
The Struggle for Diversity in Clinical Trials
The Food and Drug Administration designated 2016 as the Year of Diversity in Clinical Trials. At the time, less than 10 percent of trial participants were minorities...
ICH E6: Aspects of Risk-Based Thinking
The most prominent change to Section 5 of ICH E6 is the addition of Subsection 5.0 — Quality Management, in which ICH explains the principles of risk-based thinking in quality trial management...
Also in this issue:
- Regulatory Update
- FDA Actions