April 2001 – The CenterWatch Monthly : PDF
Product Details
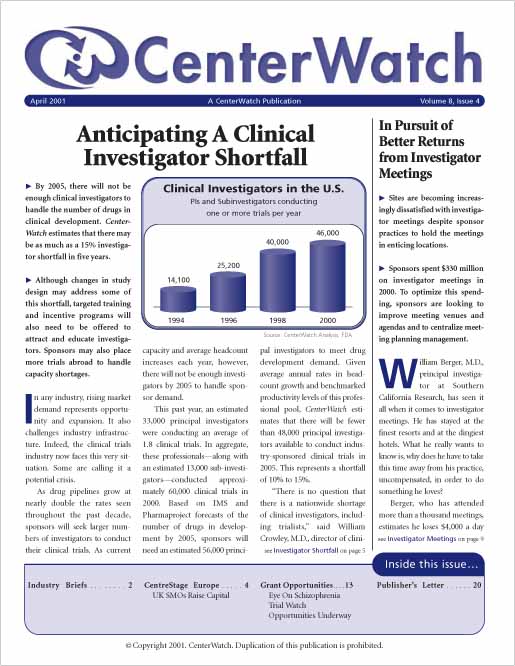
Anticipating a Clinical Investigator Shortfall
By 2005, there will not be enough clinical investigators to handle the number of drugs in clinical development. CenterWatch estimates that there may be as much as a 15% investigator shortfall in five years.
Although changes in study design may address some of this shortfall, targeted training and incentive programs will also need to be offered to attract and educate investigators. Sponsors may also place more trials abroad to handle capacity shortages.
In Pursuit of Better Returns from Investigator Meetings
Sites are becoming increasingly dissatisfied with investigator meetings despite sponsor practices to hold the meetings in enticing locations.
Sponsors spent over $330 million on investigator meetings in 2000. To optimize this spending, sponsors are looking to improve meeting venues and agendas and to centralize meeting planning management.
Also in this issue:
- CentreStage Europe: UK SMOs Raise Capital
- Eye On: Schizophrenia